3rd June, 2020
The dramatic somersault came as the pandemic carved its deadly path through Latin America, hitting hard Brazil and Peru.
The WHO has been holding clinical trials of the drug and others to find a treatment for COVID-19.
More than 380,000 people have been killed by the virus since it emerged in China last year.
In addition it has wrought massive economic damage globally.
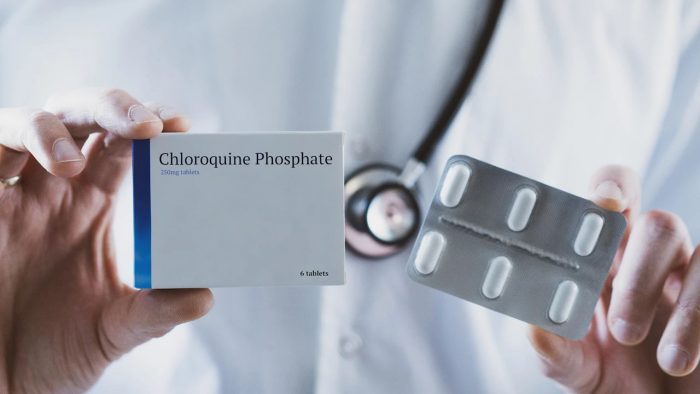
The UN agency paused trials of the hydroxychloroquine last week.
It cited a study in The Lancet medical journal that suggested it could increase the risk of death among COVID-19 patients.
But that study has come in for a steady stream of criticism.
The Lancet has issued an “expression of concern” to acknowledge the seriousness of the questions raised.
In a dramatic shift, WHO boss, Tedros Ghebreyesus Wednesday night announced that the agency will continue the trials.
“Last week the #COVID19 Solidarity Trial Executive Group implemented a temporary pause of the hydroxychloroquine arm of the trial, because of concerns about the safety of the drug”, Tedros tweeted on Wednesday.
“This decision was taken as a precaution while the safety data were reviewed.
“Based on available data, the #COVID19 Solidarity Trial Data Safety & Monitoring Committee recommended there are no reasons to modify the trial protocol.
“The Executive Group endorsed the continuation of all arms of the Trial, including the use of hydroxychloroquine”, Tedros added.